Cue Health awarded $13 million government contract to develop portable, point-of-care COVID-19 test
Biotech startup Cue Health has secured a $13 million contract from the U.S. Department of Health and Human Service’s Biomedical Advanced Research and Development Authority (BARDA), which will be used to speed the development and testing of a handheld molecular test that can detect the presence of the SARS-CoV-2 novel coronavirus that causes COVID-19.
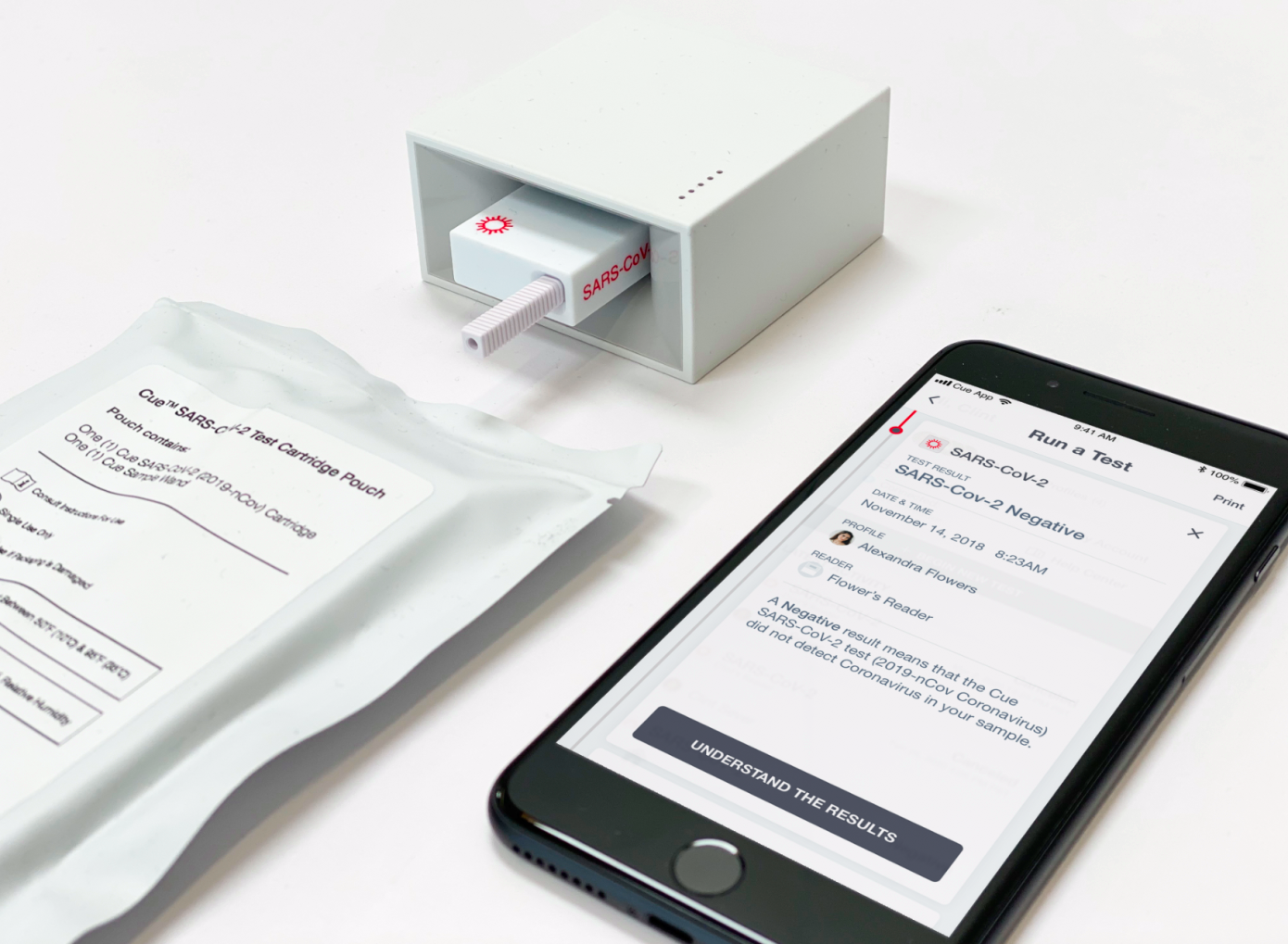
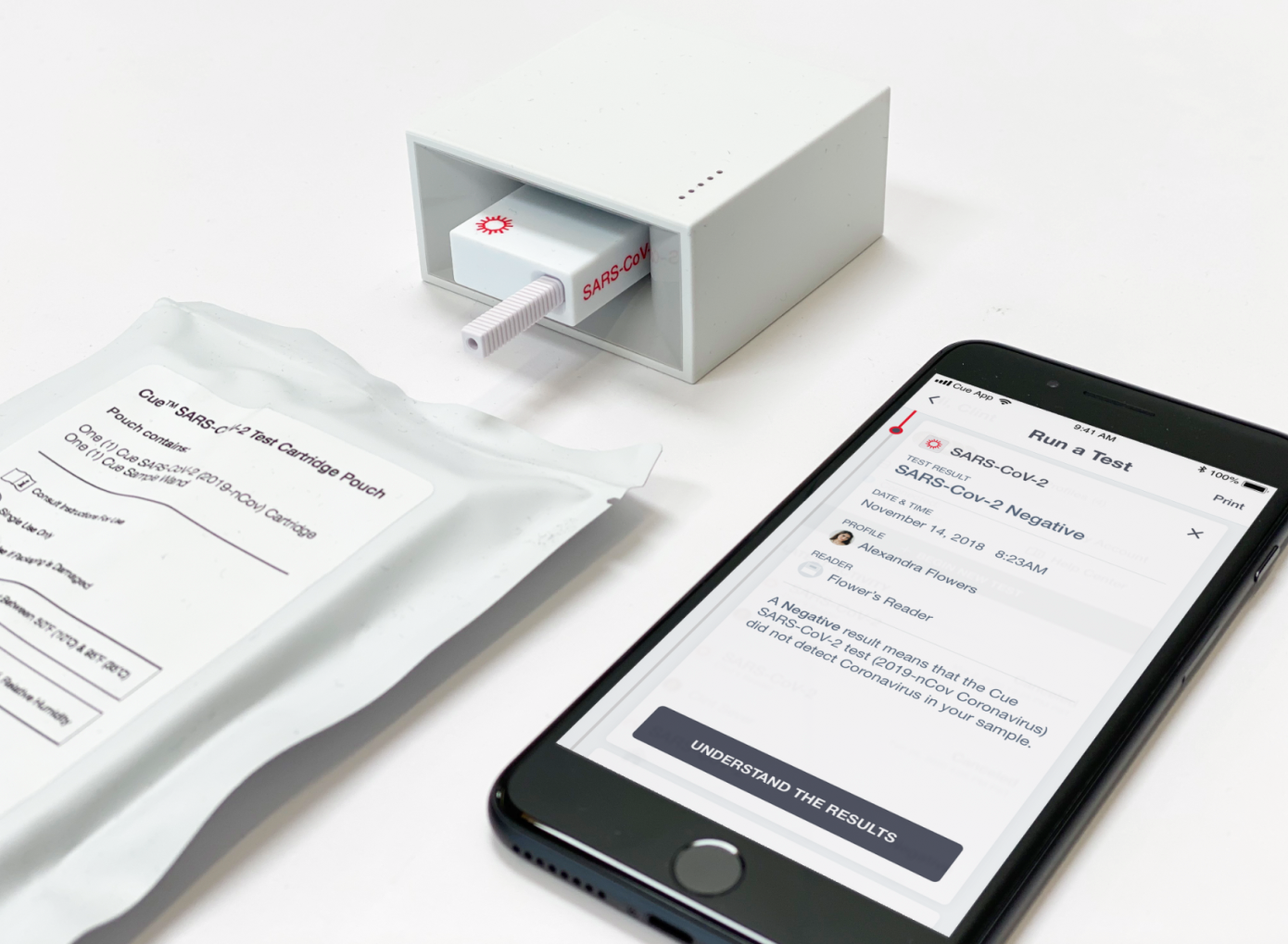
Cue, which broke cover in 2014 with plans for a connected lab in a box for at-home testing and a $7.5 million funding round, is developing a product that pairs cartridge-like test kits with a compact and connected mini lab device that can transmit results to a personalized app-based health dashboard.
The startup received a previous $30 million contract from BARDA in 2018, which was earmarked for the development and validation of an over-the-counter diagnostic test for influenza and multiplex respiratory pathogens. This pre-existing relationship and work will be useful in helping jump-start the effort on developing COVID-19 testing, the company says.
“We have worked with the BARDA team for the past two years developing and testing a 20-minute, molecular influenza test designed for home and point-of-care use,” said Cue Health CEO Ayub Khattak in a statement. “Our connected platform could serve as a critical tool in identifying the SARS-CoV-2 virus.”
The company also raised a $45 million Series B funding round the same year, which was designed to help it fund the first set of FDA clinical products used to validate its first products aimed at providing consumer diagnostics.
Cue’s proposed test solution would provide results in less than 25 minutes, using samples collected via nasal swab, with all testing done at point-of-care rather than requiring any round-trip shipping.
It’s far from the only rapid, point-of-care test either in development, in testing or already approved for use under the FDA’s Emergency Use Authorization, and there’s no specific timeline for this to become available. But the fact remains that the current testing gap needs to be addressed essentially by as many solutions as can be proven effective and viable — and this work should be useful long-term in addressing similar global crises and pandemics in the future.
Original Post: TechCrunch, 3/31/20